Description
- High-purity technical antifreeze fluid used in building engineering, heat recovery systems, and as a coolant in vehicles.
- In industrial environments, ethylene glycol is more widely used due to its lower purchase cost, lower viscosity resulting in reduced operating costs (lower pump consumption), and superior heat transfer properties.
- Ethylene glycol is toxic and classified as a hazardous substance under REACH regulations; therefore, its use requires increased attention to workplace safety.
Specification
- Freezing point: -70 °C
- Boiling point: 197.3 °C
- Density: 1.1132 g/cm³
- Viscosity: 16.1 mPa·s
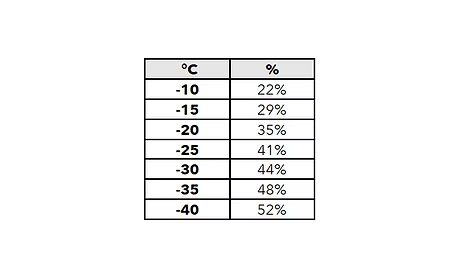
- The freezing point is not directly proportional to dilution.
- A mixture of 1 part water and 1 part ethylene glycol has a freezing point of -37 °C.
- Excellent corrosion resistance and high thermal stability.
- Recommended replacement interval: 2–3 years.
General Applications
- Chemical raw material
- Heat transfer medium
- Coolant mixtures
- De-icing agents
- Polymer fiber production
- Paint industry
- Detergent manufacturing
- Metal processing
- Agrochemicals
- Water treatment
- Lubricants
- Use of propylene glycol is not recommended in systems designed for ethylene glycol, as its higher viscosity may alter pump performance and reduce cooling efficiency.
Sustainability Summary
Production Process
Standard industrial ethylene glycol
Use
➡️
➡️
Used ethylene glycol
➡️
Collection & purification
Standard ethylene glycol
Standard ethylene glycol
↔️
Use
Used ethylene glycol
Used ethylene glycol
Life Cycle Assessment (LCA) – per 1 ton (T)
- 🌱 Based on RISE Research – Swerea database (from glycol collection to the end of the purification process)
- 🌍 Carbon footprint: ≈ 400 kg CO₂ eq/T, equivalent to approximately (US EPA data): ~1620 km driven (car), ~170 liters of gasoline consumed, ~200 kg of coal burned, ~0.5 months of energy use for an average European household, ~1 barrel of crude oil burned
- ♻️ Offset requirement: ≈ growth of 7 tree saplings over 10 years, ≈ carbon sequestration of 0.2 hectares of forest per year